Digital healthcare integrations
Digital healthcare integrations
Digital health is the amalgamation of software and hardware technology and the delivery and management of healthcare, encompassing a wide range of fields, from wearable devices to artificial intelligence, each associated with very different interaction and data collection model definitions.
Patient
Introduction
Many healthcare companies provide various terms of information related to digital health technologies for example such as digital health, connected devices, smart devices, wearables, activity trackers, connected sensors, etc. Remote monitoring, electronic surveys, electronic patient report results, telemedicine, telemedicine, artificial intelligence, chatbots, and digital assistants. To filter results for cancer-specific applications, the terms Healthcare and Cancer are added to the above terms. These results included studies that exemplify the use of digital health technologies in various areas of cancer treatment.

Connected devices
The concept of connected devices or the Internet of Things refers to devices that can transmit information remotely over an intranet or internet. The number of internet-connected devices is growing rapidly, reaching nearly 23 billion devices in 2016. This is expected to double to 50 billion devices by 2022, with a corresponding market value of about $163 billion. Connected devices include internet-connected blood pressure monitors and weight scales. Products range from smart meters to wearable activity trackers and biometrics monitoring devices. Depending on the specific application, connected devices may require and can use active, passive or hybrid data acquisition.
Wearable device systems: Passive data collection.
There is an understanding of how over the past decade interest in the integration of wearable technology into cancer care has grown, especially given its ability to integrate passive data collection and minimize the patient burden. Research on ClinicalTrials.gov using the above keywords found approximately 35 ongoing or recently completed or registered clinical trials investigating activity this tracker use in combination with cancer treatment. Wearable technology has the potential for long-term data collection allowing healthcare professionals to work better without the significant patient burden and their privacy while ensuring better-functioning integration of health system development.
A recent study demonstrated the benefits and feasibility of using a wrist-mounted activity tracker worn continuously from the initiation of radiotherapy up to 4 weeks after completion to monitor patients receiving concomitant chemoradiation therapy. Was prospectively evaluated at approximately 80% threshold. Interestingly, in exploratory analyses, step count correlated with short-term risk of hospitalization during treatment, but not with reduced physician-assessed performance status. This is suggestive of a role for continuous monitoring via wearable devices can help as a potential early-warning indicator for adverse outcomes, independent of traditional physician-reported metrics.
In addition to serving as early-warning indicators of toxicity, efforts have also been made to correlate data acquired from activity trackers with QOL (quality of life). One such study prospectively investigated patients with advanced lung cancer who were given Fitbit activity trackers to measure daily step counts and multiple validated QOL questionnaires. The authors demonstrated that higher physical activity levels (as measured by daily step count) positively correlated for example with physical functioning, role functioning, emotional functioning, and global QOL. A separate study examining patients undergoing stem cell transplantation found similar correlations, where step counts were associated with the National Cancer Institute’s Patient-Reported Outcomes–Common Terminology Criteria for Adverse Events and multiple QOL items from PROMIS (Patient-Report Outcomes Measurement Information System) Multiple other studies have been published exploring the use and feasibility of activity trackers in various subsets, such as prostate, ovarian and pediatric cancer populations.
Beyond wrist-mounted activity trackers, other novel wearable devices are also entering the healthcare domain. One such device includes a small, chest-mounted device, called ADAMM (Automated Device for Asthma Monitoring and Management), which collects multiple parameters, such as respiration rate, heart rate, body temperature, coughing/wheezing, and step-count data. This device was originally developed and validated in patients with asthma as a platform to facilitate continuous symptom monitoring in adolescents and was found to be a significant predictor of overall asthma control and healthcare utilization after 3 months. It is now being actively investigated in an open clinical trial exploring the feasibility of using this device for monitoring adult patients with lung cancer receiving active radiotherapy.
As demonstrated previously, the proliferation of wearable devices has enabled clinicians to obtain activity and biometric data with minimal part of the patient burden, with results suggesting a correlation with clinically meaningful endpoints, such as hospitalization rate and QOL. However, there remains no current standard for activity data goals at this time, and further clinical trials are warranted to explore this question. In addition, currently published studies of wearable devices, which focus primarily on data interpretation by physicians, could benefit from a focus on more clearly defined patient education. Through physician-driven biometrics and activity goals, patients can be monitored via an accompanying application health system.
Telehealth/Telemedicine
Telemedicine, which the Health Resources and Services Administration defines as “the use of electronic information and telecommunications technologies to support long-distance clinical health care, patient and professional health-related education, public health, and health administration,” has been explored over the past several decades in various forms, and in recent years, has seen augmentation by a number of new digital technologies, and the possibility of digital transformation development in future. Telehealth offers the potential for blending both audio and visual interaction with a remote patient and allows for bidirectional information flow, in contrast to the previously discussed digital survey collections. Multiple studies have demonstrated that psychosocial aspects of disease and treatment can be effectively addressed through telephone follow-up compared with in-person treatment. Studies in various cancer subgroups have demonstrated the feasibility and acceptability of conducting post-treatment follow-up visits via telemedicine, including for patients with brain, breast, prostate, endometrial, bladder, and colorectal cancer.
The proliferation and adoption of mobile technology in recent years have expanded the scope of telemedicine integration in healthcare. One notable example has been the emergence of teledermatology for skin cancer screening/triage. A randomized study investigating the utility of teledermatology was recently published, in which 484 patients were enrolled to compare the diagnostic performance of clinical teleconsultations with clinical plus Internet-based dermoscopic teleconsultations. The combination of Teledermoscopy with clinical teleconsultation resulted in an improved sensitivity and specificity of 93% and 96%, respectively, compared with an in-person dermatology consultation control group. In addition to potential cost benefits, the impact of telemedicine on managing the quality of life of cancer patients has also been investigated. One of these studies examined the role of telemedicine combined with telemonitoring of symptoms in 405 cancer patients with depression, cancer-related pain, or both. The 45 patients assigned will use an automated home symptom monitoring solution via interactive voice recording calls or web-based surveys with remote telephone support from a combined team of nurses/experts. Using this telemedicine system, patients who had depression at enrollment showed significant improvement in their depression severity scores, and patients who had cancer-related pain at enrollment showed more positive feedback to patients. It showed significant improvement in pain severity scores on the Pain Brief Inventory compared to the intervention.
Chatbots and Intelligent Assistants
Chatbots are artificial intelligence-driven software programs designed to interact with humans in a conversational fashion. Chatbots are ubiquitous in the industry and are often used as part of customer service screening. Similar technologies, coupled with more advanced artificial intelligence and neural network learning capabilities, power popular voice-based smart assistants such as Amazon’s Alexa, Google’s Assistant, Microsoft’s Cortana and Apple’s Siri. Similar to telemedicine solutions, chatbots and intelligent assistants offer the possibility of two-way information exchange with patients. Additionally, these digital assistants include text-based services (such as SMS [Short Message Service] text messages, mobile applications, or web/browser-based chat windows) and primarily voice-based services (such as the Amazon Alexa platform or Google Assistant. Conversational health care with chatbots holds much potential for digital health.
At this time, there is a paucity of published clinical trials in the oncologic field using chatbots; however, examples can be found in other healthcare settings. Recently multiple clinics have released an Amazon Alexa skill, which allows users to obtain basic first aid information by asking Alexa questions such as “How do I treat a cut?” or “How do I perform cardiopulmonary resuscitation?” In addition, Mayo Clinic piloted integration of Amazon’s Alexa-powered voice assistant to answer postdischarge questions regarding skin surgeries in the dermatology center, such as “What if I see swelling?” or “Can I take a shower?”
The potential applications of digital assistants are very exciting; however, significant hurdles currently exist in their widespread application at this time. One major limitation stems from the noncompliance of many commercially available systems with the Health Insurance Portability and Accountability Act. However, other commercial entities have emerged that offer Health Insurance Portability and Accountability Act–compliant solutions, such as Orbita and Kore.ai, which may facilitate clinical integration in future studies. In summary, digital health involves the integration of various technologies into healthcare. The integration of these techniques into clinical practice has enabled a wide range of care applications, including cancer screening, assessment of baseline status, patient management during treatment, post-treatment acute follow-up, and survival. Implementing these systems can reduce costs and workflow inefficiencies, as well as improve the overall value of healthcare, patient outcomes, and quality of life.
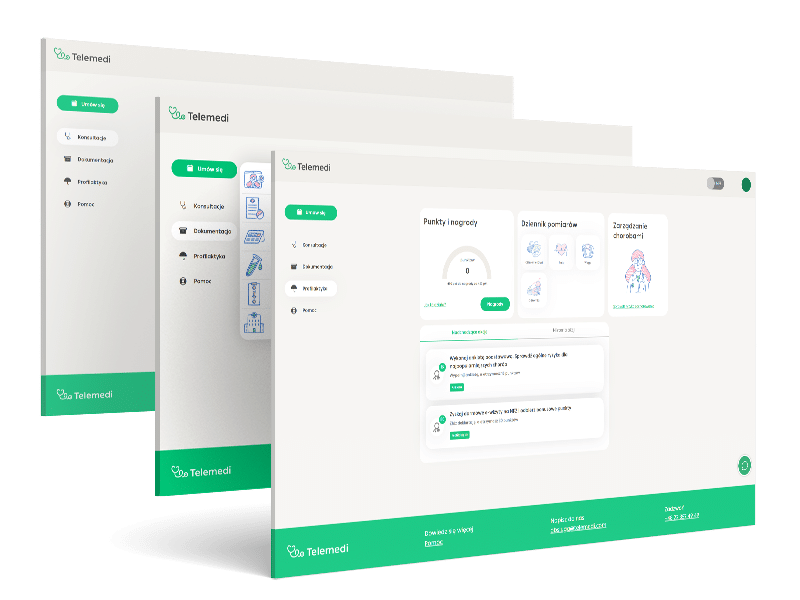
Functionalities

Dedicated accounts for the doctor, patient, and administrator

Electronic medical documentation and e-prescriptions

Collection of medical data, generation of reports, administration of system users

3 communication channels – chat, video and phone call

Platform supported by AI & Machine Learning

HIPAA & GDPR compliance
Advantages
Cost reduced by up to 60% per visit
Improved coverage in rural regions and small towns
Global coverage with various languages
Increased revenue and customers base
Improved availability to GPs and specialists, 24/7 service
Improved customers satisfaction, online consultations rated 4.8/5
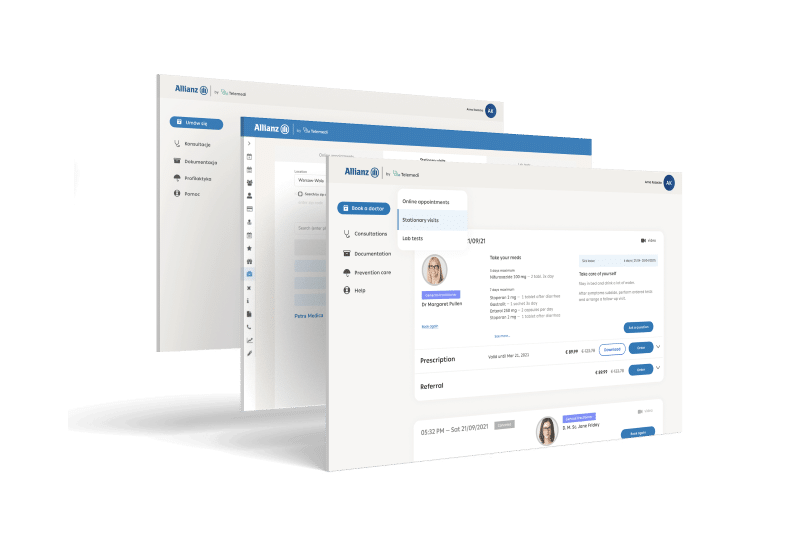
Integrated digital health solution
Telemedi platform can be used in various
business models with API integration, especially
between data, applications and devices
Let’s meet
Leave your details in the form below and lets talk!
Let’s talk about how to blend digital health with traditional healthcare efficiently.
Paweł Sieczkiewicz
CEO
Telemedi
"*" indicates required fields